
This post is the first in a series of ‘virus of the month’ articles about the viruses that the MRC-University of Glasgow Centre for Virus Research investigates. These posts are written by the CVR students and researchers and are designed to educate, engage with and inform the public and fellow scientists about the work that we are doing and the hot topics in virology.
As the ancient legend tells, the immortal Prometheus was punished by the god Zeus for providing fledgling humanity with fire. Zeus commanded that Prometheus be tied to a rock on top of a mountain where an eagle would feast on his liver each day. Yet, due to his immortality, each night when the eagle flew away, the Titan’s liver regenerated only to be devoured again come sunrise.
This punishment would go on for some thirty years until the labouring Hercules slew the eagle and freed Prometheus from his torment. The ancient Greeks certainly knew little about viruses, nor about the capacity for our liver to regenerate, but their stories continue to provide wisdom for modern-day virologists.
While it is certainly no eagle, the hepatitis C virus (HCV) continues to ravage the livers, and lives, of some 130 million people worldwide (and rising). This includes more than 200,000 in the United Kingdom. The infection can cause liver disease, which can become chronic and lead to life-threatening damage. This may even develop into liver cancer or liver failure resulting in death if a liver transplant is not performed in time. Unfortunately there is currently no vaccine available for HCV, but antiviral drugs—similar to antibiotics—may be enough to control the virus. At least, for the time being.
Developing a cure
HCV is a virus that infects your liver cells, and is spread by blood-to-blood contact, usually through non-sterile recreational or medicinal intravenous drug use. But the virus can also be caught via unprotected sex, contaminated blood transfusions or from mother to child during pregnancy. The resulting HCV hepatitis probably results from your own immune response to the virus attacking the cells it has infected.
Until very recently there were limited treatment options available for arresting an HCV infection, mainly administration of the human interferon. This is a broadly antiviral human protein, but had limited effectiveness and had to be administered for up to a year. It also had significant side-effects. It has been obvious for many years that a more effective therapy is needed.
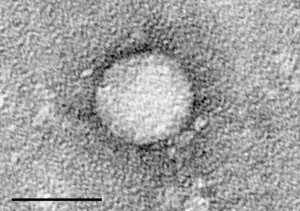
A targeted approach
HCV is roughly spherical and has a diameter of about 50 nanometers, which is 0.000000050 metres (small even for viruses). It is made up of three major kinds of molecules—its genetic material is composed of a single strand of ribonucleic acid (RNA), similar to the deoxyribonucleic acid (DNA) inside your cells; it has a hard protein shell, called a ‘capsid’, on its outer surface; and a fatty membrane, known as an ‘envelope’, studded with proteins that attach to liver cells allowing the virus to enter.
Once inside, HCV commandeers the cell’s own machinery to make hundreds of copies of itself, which can escape the cell and go on to infect nearby uninfected cells, resulting in a widespread liver infection. A cell that is forced to produce HCV, and is then targeted by an immune response, may be unable to perform its usual life-sustaining job of regulating your metabolism—helping your body break down food and removing toxins. If we made a drug that specifically prevented HCV making new versions of itself while inside your liver, it might stand a chance of curing those already infected.
Developing a drug that fights the virus, while sparing your body, had to come from understanding what makes HCV different from your own cells. HCV only makes about 11 proteins, so there is a limited choice in targets. One of the most distinct aspects of the virus is that its genetic material is composed of RNA, not DNA. Being made up of RNA requires specialised machinery or enzymes to make the new genetic copies to sustain infection. Blocking this special enzyme (called the ‘RNA-dependant RNA polymerase’), which your body lacks, should stop the infection spreading and help your body fight off the infection.
Other targets include the enzymes needed to process HCV proteins and others to replicate the viral RNA. Once discovered and characterised, drugs that interrupt these vital processes were developed and are now licensed for use in the clinic and have a remarkable ability to cure infected patients. One example is ‘Sofosbuvir’, a drug that blocks the HCV RNA polymerase, which when used with other ‘direct acting antivirals’ or treatments like interferon, can lead to a cure in up to 90% of patients. Combination therapy provides the best chance of curing a patient, and preventing antiviral resistance.
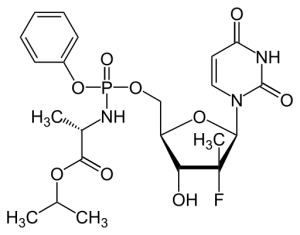
Looking to the future
Despite the great success achieved, there is a concern that some strains of the virus may prove refractory to these treatments; that the high price of the drugs might prove prohibitive to their widespread use (a course of Sofosbuvir may cost £35,000 in the UK), and that without a vaccine, HCV can never be eradicated.
Scientists across the world continue to test and refine the use of current treatments in the clinic; monitor HCV evolution and resistance within infected individuals and the community; identify and characterise new drug targets to limit infection and disease in humans; and search for a protective vaccine. Just like how Hercules aided Prometheus, society and scientists are continuing to rise to the challenge of the eagle of HCV.
The MRC-University of Glasgow Centre for Virus Research undertakes a broad research program into HCV biology ranging from epidemiological and clinical to molecular and cell biological studies. This research is led by the principal investigators: Dr Carol Leitch (HCV evolution); Dr John McLauchlan, associate director of the MRC-University of Glasgow Centre for Virus Research (HCV cell biology and immune responses, as well as being a leader of the ‘HCV Research UK’ ‘Biobank’); Dr Arvind Patel (HCV entry and vaccine development) and Dr Emma Thomson (HCV evolution and treatment in the clinic).

By Connor Bamford, Ph.D (tweets at @cggbamford)
Thanks to Dr. Naomi Bulteel (clinical research fellow working with John McLauchlan and Emma Thomson) and fellow CVR blog contributors for critical reading of this post.


[…] For scientists like me, there were very few tools to study the virus and in fact no models in the laboratory to grow HCV. So it was very much a case of building projects that still allowed us to examine the various components of the virus but not in a way that virologists would normally use. There was a lot of head scratching to try to overcome problems. Some of them still exist today even though there are now fantastic drugs that are much more effective at curing infection. […]
[…] nothing to say that what works for measles will work for any other virus. Herpes simplex and hepatitis C viruses are likely to require specialised immune […]
[…] viruses with big double-stranded DNA genomes (encoding up to 200 genes [compare that to the ten ‘genes’ of hepatitis C virus]) that infect an incredibly wide array of species including vertebrates and […]